Iron is one of the most widely used metals in the world, forming the backbone of modern civilization. From bridges and buildings to vehicles and machinery, iron plays a vital role in countless applications. But have you ever wondered, how is iron produced? This article provides a complete, step-by-step breakdown of how iron is extracted, refined, and prepared for industrial use.
Table of Contents
-
Introduction to Iron
-
Types of Iron Ore
-
The Iron Production Process
-
Mining
-
Crushing and Concentration
-
Pelletizing
-
Blast Furnace Process
-
-
Alternative Methods of Iron Production
-
Direct Reduced Iron (DRI)
-
Electric Arc Furnace (EAF)
-
-
Environmental Impact of Iron Production
-
Conclusion
1. Introduction to Iron
Iron (chemical symbol Fe) is the fourth most abundant element in the Earth’s crust. It is rarely found in its pure form; instead, it exists in ores, which are rocks containing minerals from which metals can be extracted.
The most common and economically important iron ores include:
-
Hematite (Fe₂O₃)
-
Magnetite (Fe₃O₄)
-
Limonite (FeO(OH)·nH₂O)
-
Siderite (FeCO₃)
2. Types of Iron Ore
Iron ores are rich in iron oxides and vary in color from dark grey, bright yellow, or deep purple to rusty red. Here’s a quick overview of the key types:
-
Hematite: Contains up to 70% iron, making it one of the most sought-after ores.
-
Magnetite: Slightly lower iron content than hematite but magnetic and used in specialized processes.
-
Limonite and Siderite: Lower grade ores, often used when other resources are scarce.
3. The Iron Production Process
The production of iron involves several stages, from mining to refining. Let’s take a look at each step.
A. Mining
The journey of iron begins with mining. Iron ore is extracted from open-pit or underground mines using heavy machinery. Once extracted, the raw ore is transported to processing plants.
B. Crushing and Concentration
At the plant, the ore is crushed into smaller pieces using crushers and grinders. After crushing, it undergoes concentration to increase the iron content. Methods such as magnetic separation or flotation are used to remove impurities like silica and alumina.
C. Pelletizing (Optional)
In many cases, the concentrated iron ore is formed into pellets (small balls) to improve efficiency in the blast furnace. These pellets are heated and hardened before being used in the next stage.
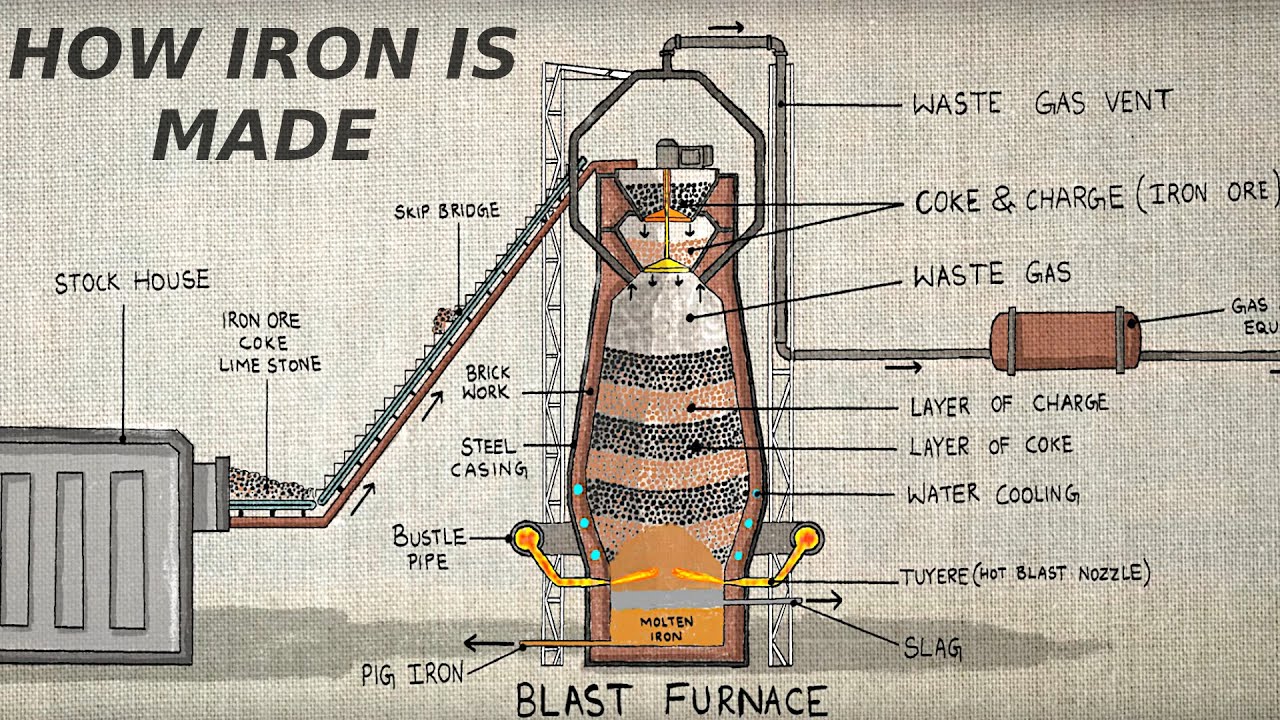
D. Blast Furnace Process
The most traditional and widely used method of producing iron is through the blast furnace.
How it works:
-
Inputs: The blast furnace is charged with iron ore, coke (a form of carbon), and limestone.
-
Combustion: Hot air is blasted into the bottom of the furnace. The coke burns to produce carbon monoxide (CO), which reacts with iron oxide to produce molten iron.
-
Chemical Reaction:
Fe2O3+3CO→2Fe+3CO2Fe₂O₃ + 3CO → 2Fe + 3CO₂
-
Slag Formation: The limestone reacts with impurities to form slag, which floats on top of the molten iron and is removed.
-
Tapping: Molten iron (called pig iron) is drained from the bottom of the furnace and transported to steelmaking facilities.
4. Alternative Methods of Iron Production
As technology and environmental concerns evolve, alternative methods have emerged.
A. Direct Reduced Iron (DRI)
Also known as sponge iron, DRI is produced by directly reducing iron ore using natural gas or coal instead of coke. The result is a solid, porous iron product.
Advantages:
-
Lower carbon emissions
-
Can be used in electric furnaces
B. Electric Arc Furnace (EAF)
EAFs are commonly used to melt scrap steel or DRI using electricity. While not a direct iron production method, they are essential in modern steel recycling and manufacturing.
Advantages:
-
Flexible in scale
-
More environmentally friendly
5. Environmental Impact of Iron Production
Iron production, especially via blast furnaces, has a significant environmental footprint.
-
Air Pollution: CO₂, NOx, SO₂ emissions
-
Water Usage: Large volumes needed for cooling and processing
-
Mining Impact: Land degradation, deforestation
Modern plants implement technologies to capture emissions, recycle water, and reclaim land, but the industry still faces sustainability challenges.
6. Conclusion
Iron production is a complex, multi-step process involving mining, refining, and smelting. While the traditional blast furnace remains the dominant method, Direct Reduced Iron and Electric Arc Furnace techniques are gaining popularity due to their lower environmental impact.
Understanding how iron is produced not only gives insight into the backbone of industrial development but also highlights the need for sustainable practices in metallurgy.